RETINA & GLAUCOMA
LIGHTLas 810
Infrared Diode Photocoagulation Laser. Deep Penetration with SP-Mode® Microsecond Laser Technology

LIGHTLas 810
Infrared Diode Photocoagulation Laser
The LIGHTLas 810 delivers 3.0W of 810nm infrared diode laser energy for deep tissue penetration. This delivers a versatile platform covering peripheral retinal photocoagulation, transscleral cyclophotocoagulation (TSCPC), and retinopathy of prematurity (ROP) treatment from a single compact console.
- Deep tissue penetration: 810nm infrared effective through cataracts, vitreous hemorrhage, and sclera.
- True Solid-State 3.0W Performance: Provides robust energy for peripheral retina and transscleral cyclophotocoagulation, while solid-state architecture ensures dependable output and long-term system durability.
- Transscleral cyclophotocoagulation: reduce IOP in refractory glaucoma with both Continuous Wave (CW) and SP-Mode® protocols.
- SP-Mode® Microsecond Laser Technology: customized protocols to fine-tune the balance of efficacy and safety for each case.
- Multiple delivery options: slit lamp, laser indirect ophthalmoscope, endoprobe, and cyclophotocoagulation probes.
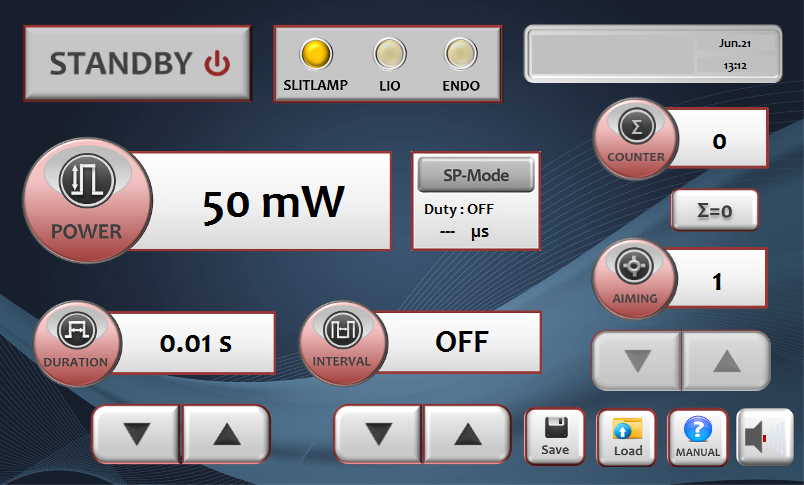
- 7-inch LCD touchscreen: intuitive interface with adjustable spot size and saveable treatment protocols.
810nm Infrared Wavelength
Infrared 810nm wavelength penetrates deeper into ocular tissues compared to visible wavelengths. The infrared characteristic enables effective photocoagulation through cataracts, vitreous hemorrhage, and corneal opacities while reducing visible light scatter and glare.
Transscleral Cyclophotocoagulation
The 810nm wavelength penetrates sclera effectively, enabling transscleral ciliary body ablation for IOP reduction in refractory glaucoma. Compatible with cyclophotocoagulation probes for both Continuous Wave (CW) and SP-Mode® treatment protocols.
High-Power 3.0W Output
True solid-state design delivers up to 3.0W continuous power for peripheral retinal photocoagulation and cyclophotocoagulation applications. The high power output ensures sufficient energy delivery even through media opacities or when treating through the sclera.
SP-Mode® Microsecond Laser Technology
Built-in sub-threshold microsecond pulsing enables tissue-sparing laser therapy. SP-Mode® with 810nm offers customized protocols which fine-tune the balance of efficacy and safety for each case, extending treatment options for advanced or complex eyes.
Multiple Delivery Options
Connect to slit lamp delivery systems, laser indirect ophthalmoscope, 20G/23G/25G endophotocoagulation probes, or transscleral cyclophotocoagulation probes. One console supports your complete infrared laser workflow across all clinical applications.
7-Inch Touchscreen Interface
Intuitive LCD touchscreen simplifies parameter adjustment for both photocoagulation and cyclophotocoagulation modes. Adjustable spot size from the console allows you to optimize treatment for different retinal zones without changing probes.
See the touchscreen interface →Combo System Availability
Available as a standalone 810nm console or in combination with the LIGHTLas SLT Deux-V or LIGHTLas YAG-V. Combo configurations let you add glaucoma SLT or YAG capsulotomy capability without a second laser platform.
See the SLT Deux-V/810 combo system →Responsive Service & Support
Backed by LIGHTMED’s dedicated service team with local service centers. Practice-friendly warranty and service plans help minimize downtime and keep your clinic running smoothly.
Accessories
- Endoprobes (straight, flexible, illuminated) 20G, 23G, 25G, 27G
- Cyclophotocoagulation Probe
- TruSpot Slit Lamp Adaptor (SLA) for Haag-Streit (analogues)
- TruSpot Slit Lamp Adaptor (SLA) for YAG-V, LIGHTLas SLT, and LIGHTLas SLT Deux-V
- Integrated Slit Lamp (SL980)
- Keeler Vantage Laser Indirect Ophthalmoscope (LIO)
- Power control foot pedal
- Motorized and fixed safety filter for microscopes
- Mobile SMART Cart
LIGHTLas 810 Technical Specifications
| Laser System | Diode Laser |
| Laser Safety Classification | Class 4 |
| Wavelength | 810 nm infrared |
| Power Output | 0.05 – 3.0 W, continuously variable |
| Max Power at Cornea | 3.0 W (Endo, LIO and SLA at all spot sizes) |
| Pulse Duration | 0.01 – 10.0 s, continuously variable |
| Pulse Interval | Variable from 0.01 – 3.0 s, and continuous |
| SP-Mode® Settings (Sub-Threshold Laser Therapy) |
Duration: 0.01 – 3.0, 3.5, 4.0, 4.5, 5.0, 10 – 90 s Duty Cycle: 5%, 7.5%, 10%, 12.5%, 15%, 20%, 25%, 30%, 31.3% Period: 0.01 – 3.0, 3.5, 4.0, 5.0, 10.0 s and OFF |
| Cooling | Auto Fan & TEC’s for Laser & Crystal |
| Treatment Spot Size | 50 – 1000 µm Integrated Version |
| Aiming Beam | Laser diode 635–650 nm red, 0.1 – <1 mW, max. power 1.0 mW |
| Slit Lamp Illumination | LED XLamp® XM-L2 2.85 V 10 W |
| Aiming Laser Safety Classification | Class 2 |
| Dimensions (Laser Console) | 13 cm (H) × 36 cm (W) × 33 cm (D) | 5.1 in (H) × 14.5 in (W) × 12.9 in (D) |
| Weight (Laser Console) | 7.8 kg | 17.2 lbs |
| Power Requirements | 100–230 VAC, 50–60 Hz Auto Ranging |
Laser Indirect Ophthalmoscope
| Indirect Model | Keeler Vantage |
| Retinal Spot Size | 1100 µm, measured at 280 mm from the front face of the LIO |
| Illumination Power | From laser console or stand alone power source |
| Fiber Length | 5 m |
| Weight | 800 g |
| Safety Filter | Fixed filter OD 4 @ 810 nm |
Specifications subject to change. Refer to the operator manual for complete technical details.
Supporting Research for 810nm Diode Laser
Infrared 810nm diode lasers are established for peripheral retinal photocoagulation and transscleral cyclophotocoagulation. Below are studies from the LIGHTMED clinical library.
Case Report: A Two-Step Approach in the Management of Angle-Closure Glaucoma Associated with Plateau Iris Configuration
Two-step approach using transscleral cyclophotocoagulation for the management of angle-closure glaucoma associated with plateau iris configuration.
Prata TS, Resende ICTP, Ribeiro KR, et al., Arq Bras Oftalmol, 2025
TESTIMONIALS