RETINA & GLAUCOMA
LIGHTLas 532
Green Photocoagulation Laser. True Solid-State 2.0W with SP-Mode® Microsecond Laser Technology

LIGHTLas 532
Green Photocoagulation Laser
The LIGHTLas 532 delivers true solid-state 2.0W of 532nm green laser energy with dual operational modes. For patients, SP-Mode® Microsecond Laser Technology means gentler, tissue-sparing treatments without visible burns, reducing discomfort and the frequency of anti-VEGF injections. For practices, it means a compact, reliable console with multiple delivery options that handles your entire retinal laser workflow from one device.
- Comprehensive CW and SP-Mode® Capability: Delivers traditional Continuous Wave photocoagulation for PRP, focal, and grid treatments, alongside SP-Mode® sub-threshold therapy for tissue-sparing care with reduced discomfort and no visible burns.
- True solid-state 2.0W at 532nm: delivering a stable, reliable green wavelength with optimal hemoglobin and melanin absorption for precise treatment and long-term system durability.
- Multiple delivery options: compatible with slit lamp, laser indirect ophthalmoscope, and endophotocoagulation probes.
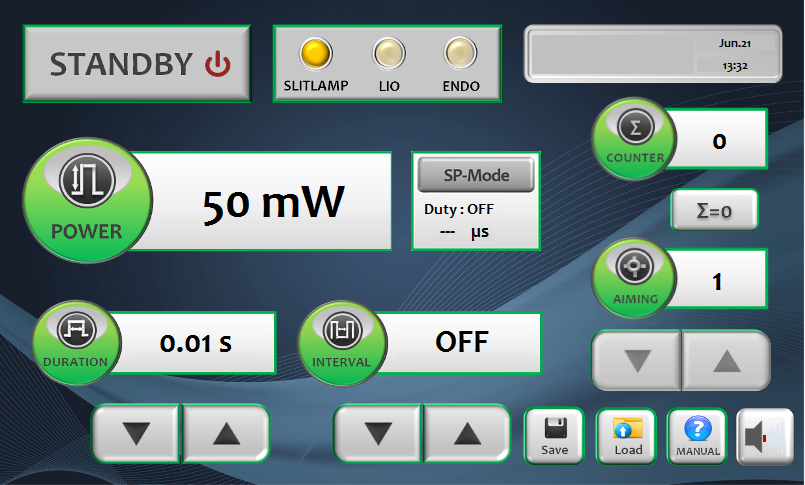
- 7-inch LCD touchscreen: intuitive interface with saveable treatment protocols for fast workflow.
532nm Green Wavelength
Green laser is preferentially absorbed by hemoglobin and melanin, making it ideal for retinal photocoagulation. The 532nm wavelength provides excellent contrast against retinal pigment epithelium while minimizing absorption in yellow macular xanthophyll pigments.
SP-Mode® Microsecond Laser Technology
Built-in sub-threshold microsecond pulsing mode enables tissue-sparing laser therapy without visible burns. SP-Mode® stimulates retinal pigment epithelium biological response while preserving photoreceptor integrity, reducing the frequency of anti-VEGF injections and improving patients’ quality of life.
Conventional Continuous Wave
Traditional Continuous Wave (CW) photocoagulation mode for panretinal photocoagulation, focal macular treatment, and grid patterns. Adjustable power (50–2000 mW) and duration (10–3000 ms) provide complete control for all standard retinal laser applications.
7-Inch Touchscreen Interface
Intuitive LCD touchscreen simplifies parameter adjustment and mode switching. Save multiple treatment protocols for different conditions so you can recall settings instantly, speeding up workflow and reducing setup time between patients.
See the touchscreen interface →Multiple Delivery Options
Connect to LaserLink integrated slit lamp, Haag-Streit adapter, laser indirect ophthalmoscope, or 20G/23G/25G endophotocoagulation probes. One console supports office-based and intraoperative workflows across all delivery modalities.
True Solid-State Design
Diode-pumped solid-state laser technology eliminates lamp replacement and reduces maintenance compared to older gas laser systems. The compact, reliable design delivers consistent 2.0W output with minimal downtime over the system’s lifetime.
Combo System Availability
Available as a standalone console or as part of a YAG-V/532 or SLT Deux-V/532 combo system. Expand your anterior and posterior segment capabilities from a single platform, maximizing your investment and simplifying your equipment footprint.
See the YAG-V/532 combo system →Responsive Service & Support
Backed by LIGHTMED’s dedicated service team with practice-friendly warranty terms and responsive local support. A better value financially with lower service costs than competing platforms.
What Sets LIGHTMED's 532nm Apart
LIGHTMED was among the earliest companies in the world to develop diode-pumped Nd:YAG → KTP 532nm green laser architecture for retinal photocoagulation. Over more than two decades, this platform has been continuously refined for stable energy output, precise beam quality, and exceptional system longevity.
While many 532nm green lasers are available today, most rely on architectures that degrade within a few years. LIGHTMED's diode-pumped solid-state design eliminates the thermal stress and internal wear common in older flashlamp and gas laser systems. The result is a laser that routinely delivers up to 10 years of reliable clinical operation.
Key Advantages
- Stable energy delivery with minimal output variation
- High beam quality with uniform circular spots and precise placement
- Reduced thermal stress on optical components for extended crystal lifetime
- Optimized optical cavity architecture and precision crystal bonding
- Intelligent system monitoring for real-time performance tracking
Accessories
- Endoprobes (straight, flexible, illuminated) 20G, 23G, 25G, 27G
- TruSpot Slit Lamp Adaptor (SLA) for Haag-Streit (analogues)
- TruSpot Slit Lamp Adaptor (SLA) for LIGHTLas YAG-V, LIGHTLas SLT, and LIGHTLas SLT Deux-V
- LaserLink Integrated Slit Lamp (SL980)
- Keeler Vantage Laser Indirect Ophthalmoscope (LIO)
- Power control foot pedal
- Motorized and fixed safety filter for microscopes
- Mobile SMART Cart
LIGHTLas 532 Technical Specifications
| Laser System | Diode Pump Solid State (DPSS) true CW and SP-Mode® |
| Laser Safety Classification | Class 4 |
| Wavelength | 532 nm green |
| Power Output | 0.05 – 2.0 W, continuously variable |
| Max Power at Cornea | 2.0 W (Endo, LIO, and SLA at all spot sizes) |
| Pulse Duration | 0.01 – 3.0 s, continuously variable |
| Pulse Interval | Variable from 0.01 – 3.0 s, and continuous |
| SP-Mode® Settings (Sub-Threshold Laser Therapy) |
Duration: 150 µs – 600 µs (in 50 µs increments) Duty cycle: 5% – 30% (in 2.5% increments) Period: 1400 µs – 1850 µs (in 50 µs increments) |
| Cooling | Auto Fan & TEC’s for Laser & Crystal |
| Treatment Spot Size | 50 – 1000 µm Integrated Version |
| Aiming Beam | Laser diode 635–650 nm red, 0.1 – <1 mW, max. power 1.0 mW |
| Slit Lamp Illumination | LED XLamp® XM-L2 2.85 V 10 W |
| Aiming Laser Safety Classification | Class 2 |
| Dimensions (Laser Console) | 13 cm (H) × 36 cm (W) × 33 cm (D) | 5.1 in (H) × 14.5 in (W) × 12.9 in (D) |
| Weight (Laser Console) | 9.4 kg | 20.72 lbs |
| Power Requirements | 100–230 VAC, 50–60 Hz Auto Ranging |
Laser Indirect Ophthalmoscope
| Indirect Model | Keeler Vantage |
| Retinal Spot Size | 1100 µm, measured at 280 mm from the front face of the LIO |
| Illumination Power | From laser console or stand alone power source |
| Fiber Length | 5 m |
| Weight | 800 g |
| Safety Filter | Fixed filter OD > 5.5 @ 532 nm |
Specifications subject to change. Refer to the operator manual for complete technical details.
Supporting Research for 532nm Photocoagulation
Green 532nm laser photocoagulation is the established standard for retinal vascular disease and macular treatments. Below are published studies relevant to the LIGHTLas 532 platform.
Retrospective study of 12 eyes treated with hybrid threshold laser (532nm at 5% duty cycle) for diabetic macular edema. Retinal thickness reduced by approximately 20% at 24 weeks with minimal scar formation and stabilized macular edema.
Marashi A, Zazo A, Annals of Medicine and Surgery, 2022
Read Paper →Comparative study evaluating intravitreal bevacizumab alone versus combined bevacizumab and macular photocoagulation for treating diabetic macular edema.
Akpolat C, Evliyaoglu F, Kurt MM, et al., Turkey Clinical J Ophthalmol, 2018
Read Paper →Sub-Threshold Frequency Doubled Nd:YAG Modified Mild Macular Grid Laser for Diffuse Diabetic Maculopathy
Study evaluating sub-threshold frequency doubled Nd:YAG modified mild macular grid laser for the treatment of diffuse diabetic maculopathy.
Ahmed SI, Fasih U, Journal of Pakistan Medical Association, 2016
Going Green: Treatment Outcome and Safety Profile of Chronic Central Serous Chorioretinopathy Treated with Subthreshold Green Laser
Study demonstrating that green 532nm subthreshold laser is a safe and effective modality for chronic CSCR with very good and stable outcomes.
Khatri A, Pradhan E, Singh S, et al., Clinical Ophthalmology, 2018
TESTIMONIALS